Galvanic corrosion between aluminum and stainless steel is a common problem that can cause significant damage to structures, equipment, and components. This electrochemical reaction occurs when these dissimilar metals come into direct contact in the presence of an electrolyte, such as moisture or saltwater. Understanding how to prevent this corrosion is essential for maintaining the integrity and longevity of your metal assemblies.
To prevent galvanic corrosion between aluminum and stainless steel, you must eliminate direct metal-to-metal contact using insulating materials like rubber gaskets or plastic spacers, apply protective coatings such as paint or anodizing, and control environmental factors like moisture. The most effective approach combines physical separation with barrier coatings and proper drainage design.
Understanding Galvanic Corrosion Between Dissimilar Metals
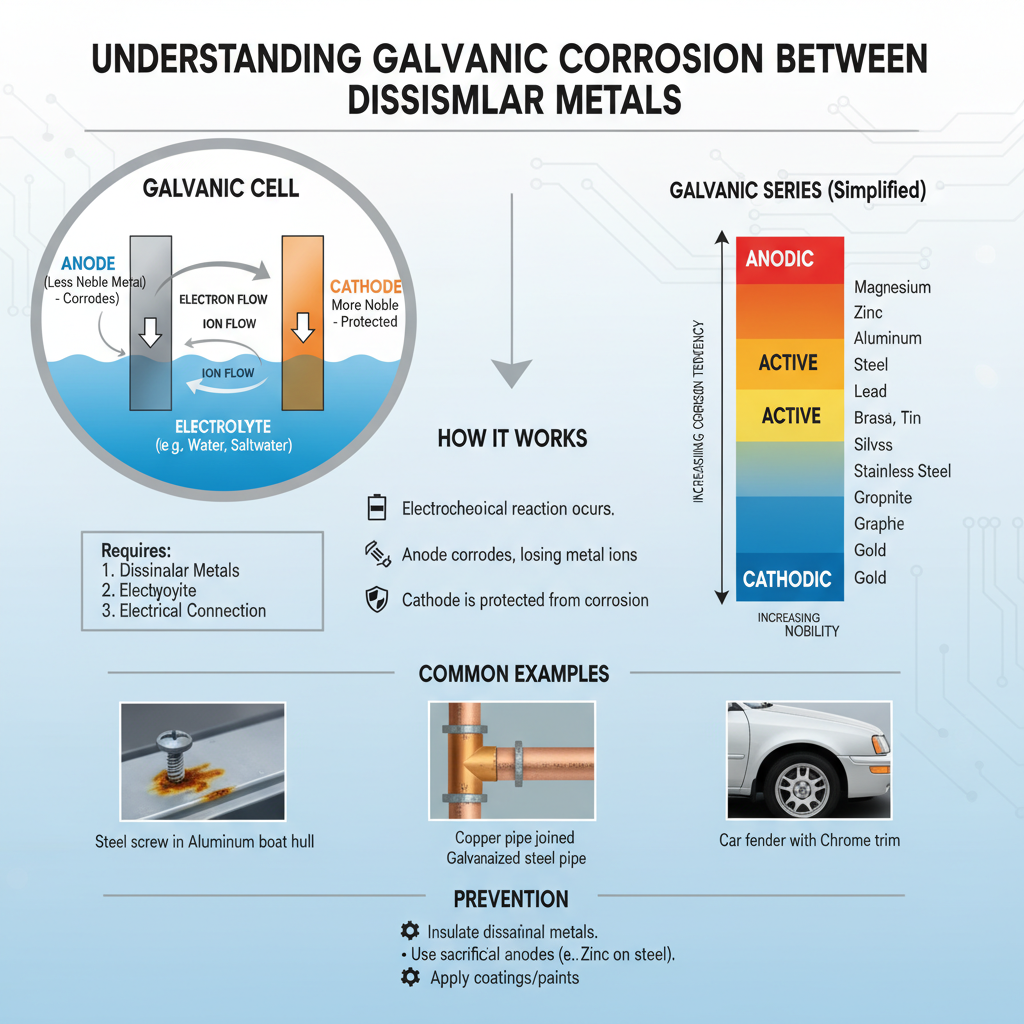
Galvanic corrosion, also known as bimetallic corrosion, happens when two different metals with varying electrochemical potentials are electrically connected in a corrosive environment. Aluminum serves as the anode (more active metal) while stainless steel acts as the cathode (less active metal). This creates an electrical current that accelerates the corrosion of aluminum.
The severity of galvanic corrosion depends on several factors:
– The difference in electrochemical potential between the metals
– The surface area ratio of the metals
– Environmental conditions (humidity, temperature, pH)
– Presence of electrolytes
Stainless steel has a much higher electrochemical potential than aluminum, making this combination particularly susceptible to galvanic corrosion. The aluminum component will corrode at an accelerated rate, potentially leading to structural failure, leaks, or complete component breakdown.
Physical Separation Methods
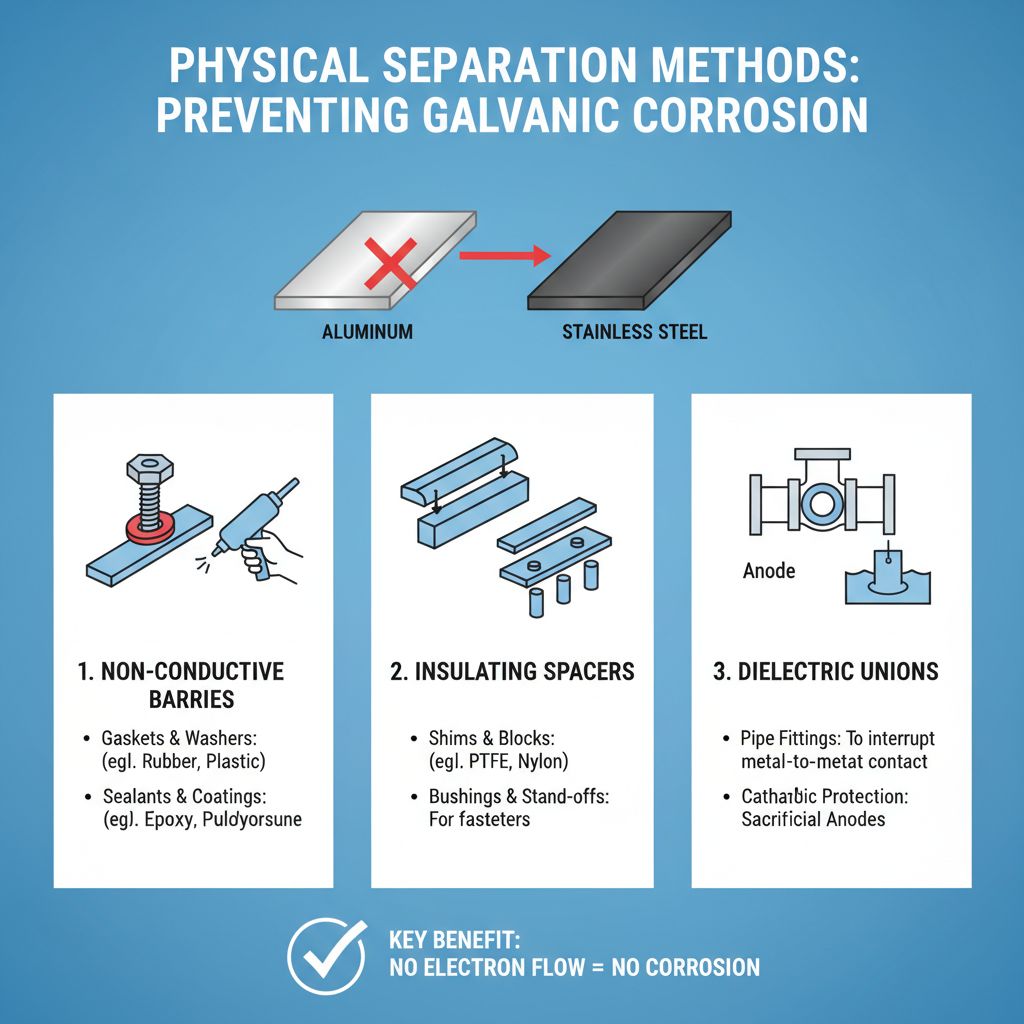
The most effective way to prevent galvanic corrosion between aluminum and stainless steel is to eliminate direct metal-to-metal contact. Physical separation creates a barrier that interrupts the electrical connection necessary for galvanic corrosion to occur.
Insulating Materials and Gaskets
Non-conductive materials serve as excellent barriers between dissimilar metals. Common insulating materials include:
– Rubber gaskets and washers
– Plastic spacers and bushings
– Nylon washers and sleeves
– Teflon tape for threaded connections
– Fiberglass reinforced plastic (FRP) components
When selecting insulating materials, consider the operating environment. Temperature extremes, chemical exposure, and UV radiation can degrade certain materials over time. Choose materials that maintain their insulating properties throughout the expected service life.
Proper Installation Techniques
Correct installation of insulating materials is crucial for long-term effectiveness. Ensure complete coverage between metal surfaces, paying special attention to bolt holes, mounting points, and contact areas. Use oversized washers or gaskets to account for thermal expansion and contraction.
Double-check that no metal-to-metal contact occurs during assembly. Even small contact points can initiate galvanic corrosion that spreads to larger areas over time.
Protective Coating Applications

Protective coatings create a barrier that prevents moisture and electrolytes from reaching the metal surfaces while also providing electrical isolation. Various coating types offer different levels of protection and durability.
Barrier Coatings
Paint systems, powder coatings, and anodizing provide effective barriers against galvanic corrosion. These coatings work by preventing the electrolyte from completing the circuit between dissimilar metals.
Zinc-rich primer offers excellent protection for both aluminum and stainless steel surfaces. This coating system provides sacrificial protection, where zinc corrodes preferentially to protect the underlying metal. Apply zinc-rich primer as a base coat followed by appropriate topcoats for enhanced durability.
For marine environments or high-humidity applications, consider epoxy-based coating systems. These coatings offer superior moisture resistance and chemical protection compared to conventional paints.
Anodizing and Conversion Coatings
Anodizing aluminum surfaces creates a thick, protective oxide layer that provides excellent corrosion resistance. This electrochemical process forms a hard, non-conductive surface that effectively isolates the aluminum from potential galvanic couples.
Conversion coatings, such as chromate or phosphate treatments, offer another layer of protection. These thin films improve paint adhesion while providing additional corrosion resistance.
Environmental Control Strategies
Controlling the environment around aluminum and stainless steel assemblies can significantly reduce galvanic corrosion rates. Since electrolytes are necessary for galvanic corrosion to occur, minimizing their presence is highly effective.
Moisture Management
Implement proper drainage systems to prevent water accumulation around metal joints and connections. Design assemblies with adequate ventilation to promote air circulation and reduce humidity levels.
Use desiccants or dehumidification systems in enclosed spaces to maintain low moisture levels. Regular inspection and maintenance of seals and gaskets prevent moisture intrusion over time.
Contamination Prevention
Salt spray, industrial chemicals, and other contaminants accelerate galvanic corrosion. Regular cleaning removes these deposits before they can cause significant damage.
Establish cleaning schedules based on environmental exposure levels. Marine environments require more frequent cleaning than indoor applications. Use appropriate cleaning solutions that won’t damage protective coatings or introduce additional contaminants.
Design Considerations and Best Practices
Proper design significantly impacts the likelihood and severity of galvanic corrosion between aluminum and stainless steel. Consider these factors during the planning and design phases.
Metal Selection and Compatibility
When possible, select metals with similar electrochemical potentials to minimize galvanic corrosion risk. If dissimilar metals are necessary, choose combinations with smaller potential differences.
Consider using aluminum alloys with improved corrosion resistance, such as 5000 or 6000 series alloys. These alloys contain elements that enhance their natural corrosion resistance.
Surface Area Ratios
The ratio of cathode to anode surface area significantly affects corrosion rates. Large stainless steel surfaces coupled with small aluminum components create severe corrosion conditions. Minimize this ratio by reducing the stainless steel surface area or increasing the aluminum surface area when possible.
Drainage and Ventilation Design
Design assemblies to promote water drainage and air circulation. Avoid creating pockets or crevices where moisture can accumulate. Include drain holes, weep holes, or other features that allow water to escape quickly.
Position sensitive joints and connections away from areas prone to water accumulation or splash zones. Elevate components above potential flood levels when practical.
Maintenance and Monitoring Programs
Regular maintenance and monitoring help identify potential galvanic corrosion issues before they become serious problems. Establish inspection schedules based on environmental conditions and criticality of the application.
Inspection Procedures
Visual inspections should focus on:
– Signs of white, powdery corrosion products on aluminum surfaces
– Pitting or localized corrosion around joints and connections
– Degradation of protective coatings
– Moisture accumulation or poor drainage
– Loose or damaged insulating materials
Use non-destructive testing methods such as ultrasonic thickness measurements to monitor metal loss over time. Document findings and track corrosion rates to predict maintenance needs.
Preventive Maintenance Tasks
Regular maintenance activities include:
– Cleaning contaminated surfaces
– Reapplying protective coatings as needed
– Replacing degraded gaskets and seals
– Improving drainage systems
– Updating insulation materials
Keep detailed maintenance records to identify recurring problems and optimize prevention strategies. Track the effectiveness of different protection methods to refine your approach over time.
Conclusion
Preventing galvanic corrosion between aluminum and stainless steel requires a comprehensive approach combining physical separation, protective coatings, environmental control, and proper design practices. The most effective strategy typically involves multiple prevention methods working together to eliminate the conditions necessary for galvanic corrosion.
Physical separation using insulating materials provides the most reliable protection by eliminating direct metal contact. Protective coatings offer additional barriers against moisture and electrolytes. Environmental control reduces the severity of corrosive conditions, while proper design minimizes risk factors.
Regular maintenance and monitoring ensure that prevention measures remain effective throughout the service life of your equipment. By implementing these strategies systematically, you can significantly extend the life of aluminum and stainless steel assemblies while avoiding costly repairs and replacements due to galvanic corrosion damage.
